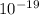Chemistry, 07.05.2020 06:06 reneecoutinho10
The addition of 5.0x10–3 total moles of Zn2+ to a 1.0 L solution of NaCN gives a solution of the complex ion [Zn(CN)4] 2– (Kf = 4.17x1019). What is the equilibrium concentration of uncomplexed Zn2+ ions if the concentration of cyanide ions at equilibrium is 0.50 M?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:30
Which of the following signs of a chemical reaction are observed in the reaction of potassium with water? precipitate formed temperature change smell produced gas produced color change
Answers: 2

Chemistry, 22.06.2019 08:30
In the reaction between a crushed antacid tablet and vinegar what gas is emitted
Answers: 2


Chemistry, 23.06.2019 01:00
Aman applies a force of 500n to push a truck 100m down the street how much does he do?
Answers: 1
You know the right answer?
The addition of 5.0x10–3 total moles of Zn2+ to a 1.0 L solution of NaCN gives a solution of the com...
Questions



English, 13.09.2019 22:30

Social Studies, 13.09.2019 22:30

Biology, 13.09.2019 22:30




Geography, 13.09.2019 22:30

Engineering, 13.09.2019 22:30

Engineering, 13.09.2019 22:30


Engineering, 13.09.2019 22:30







Engineering, 13.09.2019 22:30

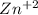 = 1.9 *
= 1.9 *