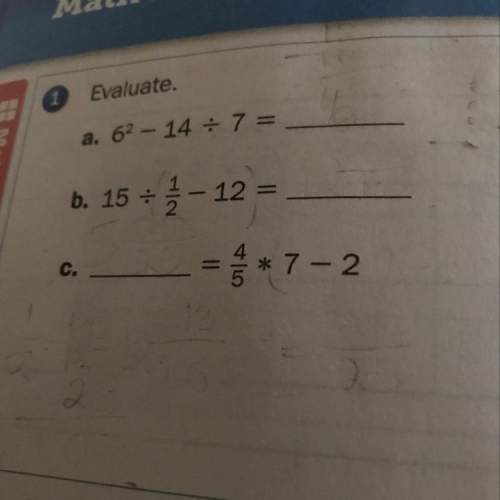Mathematics, 14.10.2020 14:01 rachael382
Calculate the ratio of H+ ions to OH– ions at a pH = 6. Find the concentration of H+ ions to OH– ions listed in Table B of your Student Guide. Then divide the H+ concentration by the OH– concentration. Record this calculated ratio in Table A of your Student Guide. Compare your approximated and calculated ratios of H+ ions to OH– ions at a pH = 6. Are they the same? Why or why not? Record your explanation in Table A. What is the concentration of H+ ions at a pH = 6? mol/L What is the concentration of OH– ions at a pH = 6? mol/L What is the ratio of H+ ions to OH– ions at a pH = 6? :1

Answers: 2
Another question on Mathematics

Mathematics, 21.06.2019 15:20
Which is the equation of an ellipse with directrices at x = ±4 and foci at (2, 0) and (−2, 0)?
Answers: 3

Mathematics, 21.06.2019 16:40
Ajar contains a mixture of 20 black marbles, 16 red marbles, and 4 white marbles, all the same size. find the probability of drawing a white or red marble on the first draw. 125 12 01 next question ask for turn it in
Answers: 2

Mathematics, 21.06.2019 20:10
Look at the hyperbola graphed below. the hyperbola gets very close to the red lines on the graph, but it never touches them. which term describes each of the red lines? o o o o a. asymptote b. directrix c. focus d. axis
Answers: 3

You know the right answer?
Calculate the ratio of H+ ions to OH– ions at a pH = 6. Find the concentration of H+ ions to OH– ion...
Questions

Mathematics, 02.11.2020 17:20


Mathematics, 02.11.2020 17:20



Mathematics, 02.11.2020 17:20

Mathematics, 02.11.2020 17:20

Spanish, 02.11.2020 17:20






World Languages, 02.11.2020 17:20


History, 02.11.2020 17:20

Mathematics, 02.11.2020 17:20