
Chemistry, 16.07.2021 23:30 Mayjay2827
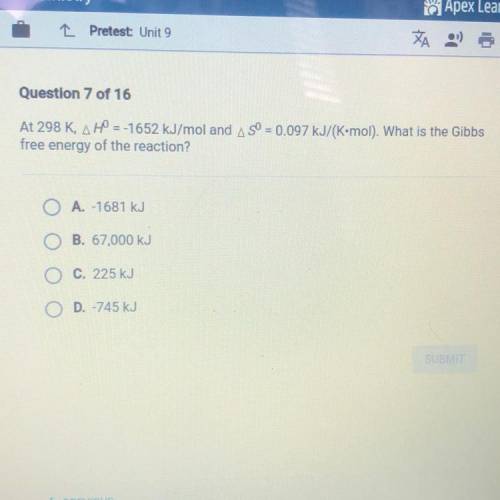
At 298 K, H = -1652 kJ/mol and S = 0.097 kJ/(K•mol). What is the Gibbs free energy of the reaction?
A.-1681 kJ
B. 67,000 kJ
C. 225 kJ
D. -745 kJ

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:50
Select the correct answer how does the heat content of the reaction change in the process of photosynthesis when a glucose molecule is formed? ca the value of is negative the value of qis positive the value of a remains constant the value of a decreases the value of equals zero e
Answers: 2

Chemistry, 22.06.2019 05:30
You are making a solution of calcium chloride dissolved in water. you add solid, stir, and it dissolves. you add just a spatula tip full, stir, and the solid does not dissolve. how could you describe the solutions before and after adding the spatula tip amount
Answers: 1

Chemistry, 22.06.2019 08:00
If 90.0 grams of ethane reacted with excess chlorine,how many grams of dicarbon hexachloride would form
Answers: 1

You know the right answer?
At 298 K, H = -1652 kJ/mol and S = 0.097 kJ/(K•mol). What is the Gibbs free energy of the reaction?...
Questions





Chemistry, 17.03.2020 00:44


Mathematics, 17.03.2020 00:44

Mathematics, 17.03.2020 00:44


Chemistry, 17.03.2020 00:44



Mathematics, 17.03.2020 00:44



Mathematics, 17.03.2020 00:44


Mathematics, 17.03.2020 00:44





