
The overall energy involved in the formation of CsCl from Cs(s) and Cl2(g) is −443 kJ mol−1. Given the following information: heat of sublimation for Cs is +76 kJ mol−1, bond dissociation energy for 12Cl2 is +121 kJ mol−1, Ei1 for Cs is +376 kJ mol−1, and Eea for Cl(g) is 349 kJ mol−1. what is the magnitude of the lattice energy for CsCl?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 08:30
What is the independent variable in this investigation? mass volume sample number substance density
Answers: 3

Chemistry, 22.06.2019 14:00
Ascientist measures the speed of sound in a monatomic gas to be 449 m/s at 20∘c. what is the molar mass of this gas?
Answers: 2

Chemistry, 22.06.2019 20:30
A40 kilogram skier starts at the top of a 12 meter high slope. at the bottom, she is travelling 10 meters per second. how much energy does she lose to friction
Answers: 2

You know the right answer?
The overall energy involved in the formation of CsCl from Cs(s) and Cl2(g) is −443 kJ mol−1. Given t...
Questions

History, 22.10.2021 20:20

Mathematics, 22.10.2021 20:20

Mathematics, 22.10.2021 20:20



Biology, 22.10.2021 20:20

Mathematics, 22.10.2021 20:20


Computers and Technology, 22.10.2021 20:20


Spanish, 22.10.2021 20:20

History, 22.10.2021 20:20



Mathematics, 22.10.2021 20:20


Mathematics, 22.10.2021 20:20


English, 22.10.2021 20:20

World Languages, 22.10.2021 20:30

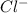 + lattice energy
+ lattice energy

