Chemistry, 31.05.2021 15:20 purplefive85
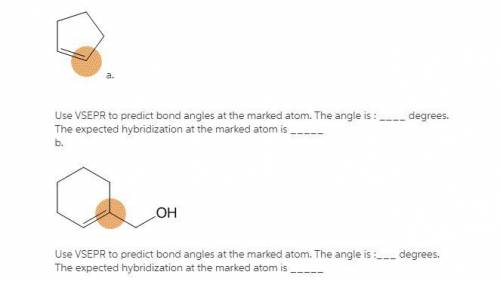
a. Use VSEPR to predict bond angles at the marked atom. The angle is : degrees. The expected hybridization at the marked atom is . b. Use VSEPR to predict bond angles at the marked atom. The angle is : degrees. The expected hybridization at the marked atom is .

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:00
If a plot weight (in g) vs. volume (in ml) for a metal gave the equation y= 13.41x and r^2=0.9981 what is the density of the metal?
Answers: 2


Chemistry, 22.06.2019 09:20
Which of these statements explains the difference between nuclear binding energy and the strong nuclear force ?
Answers: 3

You know the right answer?
a. Use VSEPR to predict bond angles at the marked atom. The angle is : degrees. The expected hybrid...
Questions


Mathematics, 12.03.2020 07:38


English, 12.03.2020 07:38


Mathematics, 12.03.2020 07:39


Mathematics, 12.03.2020 07:39

Physics, 12.03.2020 07:40

Mathematics, 12.03.2020 07:40

Spanish, 12.03.2020 07:40

Mathematics, 12.03.2020 07:40

Mathematics, 12.03.2020 07:41

History, 12.03.2020 07:41



Mathematics, 12.03.2020 07:42

Mathematics, 12.03.2020 07:43

Social Studies, 12.03.2020 07:43

History, 12.03.2020 07:43