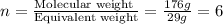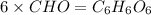Chemistry, 19.12.2020 16:10 amandaestevez030
Burning 12.00 g of an oxoacid produces 17.95 g of carbon dioxide and 4.87 g of water. Consider that 0.25 moles of oxoacid equals 44.0 g. For this compound, determine the empirical and molecular formula.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Match the following items. 1. high-intensity bundle of energy being emitted from some decaying nuclei gamma ray 2. particle radiating from the nucleus of some atoms beta particle 3. negative particle identical to an electron but radiating from a decaying nucleus alpha particle
Answers: 1

Chemistry, 22.06.2019 16:00
No copying 15 pts how does a free-body diagram tell you about the net force on an object?
Answers: 2

Chemistry, 22.06.2019 18:10
Consider the following reaction at equilibrium: c(s)+h2o(g)⇌co(g)+h2(g) predict whether the reaction will shift left, shift right, or remain unchanged upon each of the following disturbances. a) c is added to the reaction mixture. b) h2ois condensed and removed from the reaction mixture c) co is added to the reaction mixture d) h2 is removed from the reaction mixture.
Answers: 3

You know the right answer?
Burning 12.00 g of an oxoacid produces 17.95 g of carbon dioxide and 4.87 g of water. Consider that...
Questions

Biology, 10.04.2020 08:10

Mathematics, 10.04.2020 08:10

Biology, 10.04.2020 08:10

Mathematics, 10.04.2020 08:10

English, 10.04.2020 08:11







English, 10.04.2020 08:12



Mathematics, 10.04.2020 08:13



Mathematics, 10.04.2020 08:15

Mathematics, 10.04.2020 08:16

Chemistry, 10.04.2020 08:16


 = 17.95 g
= 17.95 g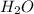 = 4.87 g
= 4.87 g of carbon will be contained.
of carbon will be contained.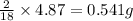 of hydrogen will be contained.
of hydrogen will be contained.

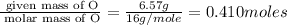



 .
.