
Chemistry, 13.02.2020 18:57 jamilamiller200
When an antacid tablet dissolves in water, the fizz is due to a reaction between sodium hydrogen carbonate (sodium bicarbonate, NaHCO3) and citric acid (H3C6H5O7). 3 NaHCO3(aq) H3C6H5O7(aq) 3 CO2(g) 3 H2O(l) Na3C6H5O7(aq) How many moles of Na3C6H5O7 can be produced if one tablet containing 0.0370 mol of NaHCO3 is dissolved

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 01:00
Which type of orbits are found in the principal energy level n = 2 a - s b - s, f c - s, d d - s, p e - s, p, d
Answers: 1

Chemistry, 22.06.2019 13:30
What are the chemical names of these compounds? ke: mg3n2: reset next
Answers: 1

Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 1
You know the right answer?
When an antacid tablet dissolves in water, the fizz is due to a reaction between sodium hydrogen car...
Questions



Mathematics, 25.01.2021 04:40





Mathematics, 25.01.2021 04:50

Mathematics, 25.01.2021 04:50





Mathematics, 25.01.2021 04:50





Mathematics, 25.01.2021 04:50

Mathematics, 25.01.2021 04:50

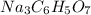 produced can be, 0.0123 mole
produced can be, 0.0123 mole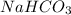 = 0.0370 mol
= 0.0370 mol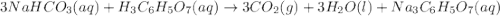
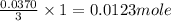 of
of 

