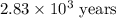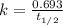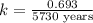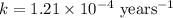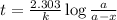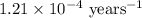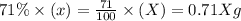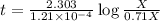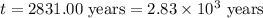Chemistry, 07.10.2019 17:30 animationfusion
Carbon-14 is a radioactive isotope that decays according to first-order kinetics in a process that has a half-life of 5730 years. if a sample containing carbon-14 now has 71% of its original concentration of carbon-14, how much time has passed in years? 4.09 ~ 103 years 5.73 x 103 years 2.38 x 103 years 2.83 x 103 years 3.52 * 104 years

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 23:30
How many grams of ammonia would be produced by the decomposition of 16.93 mlof hydrazine? (the density of hydrazine is 1.021g/ml)
Answers: 3

Chemistry, 23.06.2019 00:00
How many peaks will be present in a mass spectrum for brcl?
Answers: 1

You know the right answer?
Carbon-14 is a radioactive isotope that decays according to first-order kinetics in a process that h...
Questions

Computers and Technology, 31.12.2019 02:31




Social Studies, 31.12.2019 02:31