
Chemistry, 12.08.2019 22:30 lovecats12
Consider the following redox equation mn(oh)2(s) + mno4 –(aq) mno42 –(aq) (basic solution) when the equation is balanced with smallest whole number coefficients, what is the coefficient for oh –(aq) and on which side of the equation is oh –(aq) present?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 14:40
Water ionizes by the equation h2o(l)⇌h+(aq)+oh−(aq) the extent of the reaction is small in pure water and dilute aqueous solutions. this reaction creates the following relationship between [h+] and [oh−]: kw=[h+][oh−] keep in mind that, like all equilibrium constants, the value of kw changes with temperature.
Answers: 1

Chemistry, 22.06.2019 02:30
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2

Chemistry, 22.06.2019 11:00
The diagram below shows the different phase transitions that occur in matter. which arrow represents the transition in which dew is formed?
Answers: 1

Chemistry, 22.06.2019 12:30
According to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? view available hint(s) according to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? trigonal bipyramidal tetrahedral square planar determination of electron geometry requires information on whether the electron groups are lone pairs or bonding groups.
Answers: 2
You know the right answer?
Consider the following redox equation mn(oh)2(s) + mno4 –(aq) mno42 –(aq) (basic solution) when th...
Questions


Computers and Technology, 02.08.2019 19:30









Mathematics, 02.08.2019 19:30



Social Studies, 02.08.2019 19:30

Mathematics, 02.08.2019 19:30

Biology, 02.08.2019 19:30

Chemistry, 02.08.2019 19:30


Computers and Technology, 02.08.2019 19:30

History, 02.08.2019 19:30

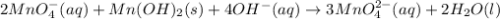
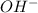 is, 4 and on reactant side of the equation
is, 4 and on reactant side of the equation 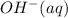 is present.
is present.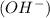 at that side where the less number of hydrogen are present.Now balance the charge.
at that side where the less number of hydrogen are present.Now balance the charge.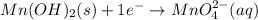 ......(1)
......(1)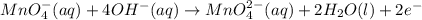 .....(2)
.....(2)

