Chemistry, 25.07.2019 02:30 brittanyfox411
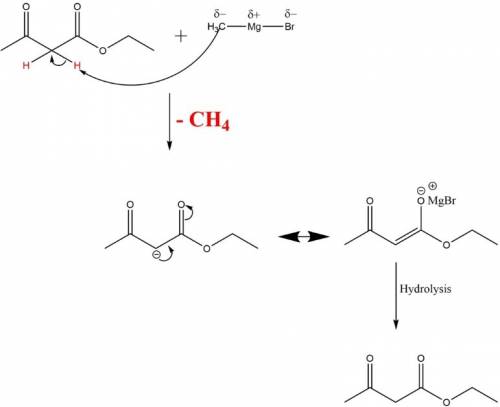
When ethyl acetoacetate (ch3coch2co2ch2ch3) is treated with one equivalent of ch3mgbr, a gas is evolved from the reaction mixture, and after adding aqueous acid, ethyl acetoacetate is recovered in high yield. identify the gas formed and explain why the starting material was recovered in this reaction. be sure to answer all parts. the gas formed is: o2 h2 ch4 co2 this occurs because: addition of a methyl group through nucleophilic addition destabilizes the ester group. because of the acidic proton, ch3mgbr reacts as a base and proton transfer occurs rather than nucleophilic addition. aqueous acid adds an oxygen to the carbanion formed releasing h2 gas. addition of aqueous acid disrupts the anhydride bond which reforms after decarboxylation?

Answers: 1
Another question on Chemistry



Chemistry, 22.06.2019 18:10
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1

Chemistry, 22.06.2019 19:30
If 16.00g of hydrogen gas reacts with 126.73g of oxygen, how many grams of water are yielded? (both reactants are completely consumed in the reaction.)
Answers: 2
You know the right answer?
When ethyl acetoacetate (ch3coch2co2ch2ch3) is treated with one equivalent of ch3mgbr, a gas is evol...
Questions

Spanish, 24.07.2019 09:20


Mathematics, 24.07.2019 09:20






Mathematics, 24.07.2019 09:20





Spanish, 24.07.2019 09:20

Mathematics, 24.07.2019 09:20





English, 24.07.2019 09:20