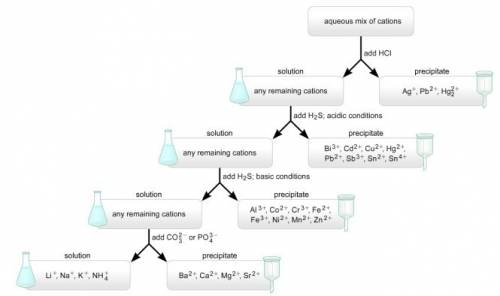
Asolution containing a mixture of metal cations was treated with dilute hcl and no precipitate formed. next, h2s was bubbled through the acidic solution. a precipitate formed and was filtered off. then, the ph was raised to about 8 and h2s was again bubbled through the solution. a precipitate again formed and was filtered off. finally, the solution was treated with a sodium carbonate solution, which resulted in no precipitation. which metal ions were definitely present, which were definitely absent, and which may or may not have been present in the original mixture?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 15:00
Many ionic compounds and a few highly polar covalent compounds are because they completely ionize in water to create a solution filled with charged ions that can conduct an electric current.
Answers: 1

Chemistry, 22.06.2019 20:50
What is the vapor pressure of a solution with a benzene to octane?
Answers: 2

Chemistry, 23.06.2019 00:00
Mercury turns to a vapor at 629.88 k. how much heat is lost when 75.0 g of mercury vapor at 650 k condenses to a liquid at 297 k?
Answers: 1

Chemistry, 23.06.2019 01:00
Who examines and coordinates the cleanup of polluted sites?
Answers: 2
You know the right answer?
Asolution containing a mixture of metal cations was treated with dilute hcl and no precipitate forme...
Questions











English, 12.03.2021 15:00