Chemistry, 28.07.2019 01:30 mallyosburn
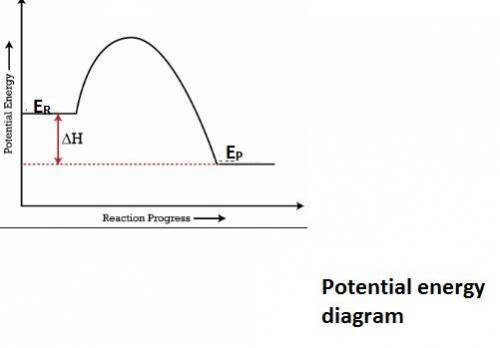
In an exothermic reaction, the potential energy of the products is lower than that of the reactants. which of the following is also true for an exothermic reaction? the enthalpy change is negative it requires a catalyst to take place energy is absorbed from the surroundings the kinetic energy of the products is also lower

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:30
What is the mass defect of a mole of nuclei with 1.8 x 10^15 j/mol binding energy?
Answers: 1

Chemistry, 22.06.2019 10:50
Someone offer some answers to this, i will give 98 coins and mark as brainliest! i will put the rest of the lab down in the comments,solutions pre-lab questions: in this lab, you will make fruit drinks with powdered drink mix. complete the pre-lab questions to get the values you need for your drink solutions. calculate the molar mass of powered fruit drink mix, made from sucrose (c12h22o11).using stoichiometry, determine the mass of powdered drink mix needed to make a 1.0 m solution of 100 ml. (hint: use molarity = to find the moles of drink mix, then convert moles to grams using a mole conversion.)what mass of powdered drink mix is needed to make a 0.5 m solution of 100 ml?
Answers: 1

Chemistry, 22.06.2019 12:30
Avariable that is not being directly tested during an experiment should be changed varied experimented controlled
Answers: 1

Chemistry, 22.06.2019 13:00
12. calculate the hydroxide ion concentration of a solution with ph = 3.25. show all calculations leading to an answer
Answers: 3
You know the right answer?
In an exothermic reaction, the potential energy of the products is lower than that of the reactants....
Questions

History, 08.10.2019 02:00


English, 08.10.2019 02:00

Mathematics, 08.10.2019 02:00


Health, 08.10.2019 02:00





English, 08.10.2019 02:00









 for the reaction comes out to be positive.
for the reaction comes out to be positive.