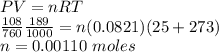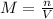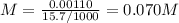The volume of a sample of pure hcl gas was 189 ml at 25°c and 108 mmhg. it was completely dissolved in about 60 ml of water and titrated with an naoh solution; 15.7 ml of the naoh solution were required to neutralize the hcl. calculate the molarity of the naoh solution.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 14:30
What state of matter is ice a. liquid b. element c. solid d. gas
Answers: 1

Chemistry, 22.06.2019 18:00
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 3

Chemistry, 22.06.2019 20:20
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3
You know the right answer?
The volume of a sample of pure hcl gas was 189 ml at 25°c and 108 mmhg. it was completely dissolved...
Questions

Mathematics, 20.11.2020 22:10


Mathematics, 20.11.2020 22:10

Mathematics, 20.11.2020 22:10

Mathematics, 20.11.2020 22:10


Mathematics, 20.11.2020 22:10


Computers and Technology, 20.11.2020 22:10




Geography, 20.11.2020 22:10

Health, 20.11.2020 22:10


Mathematics, 20.11.2020 22:10



Mathematics, 20.11.2020 22:10