
Me i beg you
the average kinetic energy of 1 mole of a gas at -32 degrees celsius is:
3.01 x 103 j
3.99 x 102 j
-3.99 x 102 j
3.80 x 103 j
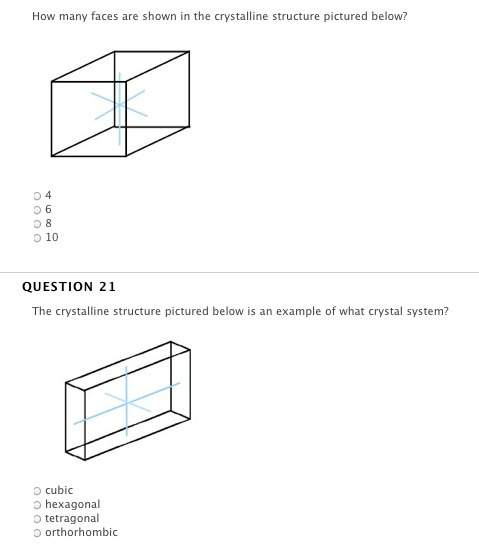
the relationship between volume and temperature of a gas, when pressure and moles of a gas are held constant, is: v*t = k.
true
false
the relationship between moles and volume, when pressure and temperature of a gas are held constant, is: v/n = k. we could say then, that:
if the moles of gas are tripled, the volume must also triple.
if the moles of gas are halved, the volume must be doubled.
if the moles of gas are doubled, the volume must be halved.
if the moles of gas are doubled, the volume is unaffected.
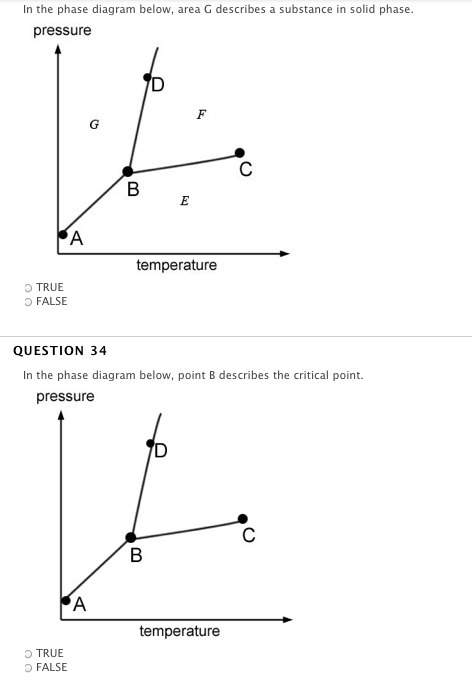
if the temperature and volume of a gas are held constant, an increase in pressure would most likely be caused by an increase in the number of moles of gas.
true
false
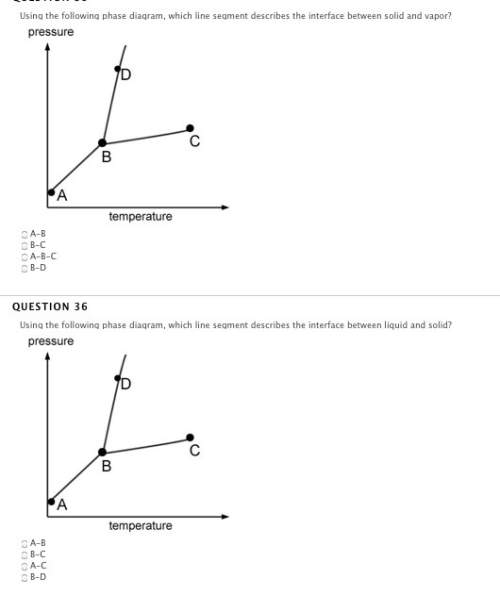
if the vapor pressure of a liquid is less than the atmospheric pressure, the liquid will not boil.
true
false


Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 12:30
The melting point of sulfur is 115 °c and its boiling point is 445 °c. what state would sulfur be in at 200 °c?
Answers: 1

Chemistry, 22.06.2019 12:50
The number at the end of an isotope’s name is the number.
Answers: 1

Chemistry, 22.06.2019 15:00
Many ionic compounds and a few highly polar covalent compounds are because they completely ionize in water to create a solution filled with charged ions that can conduct an electric current.
Answers: 1

Chemistry, 22.06.2019 15:30
The gulf stream is a warm water current that flows away from the equator to northern europe. witch of these does it cause. a. crashes of warm and cool water in the ocean b.colder climates near the equator c.large waves on the cost of europe d.warm climates in northern europe
Answers: 1
You know the right answer?
Me i beg you
the average kinetic energy of 1 mole of a gas at -32 degrees celsius is: <...
the average kinetic energy of 1 mole of a gas at -32 degrees celsius is: <...
Questions



Mathematics, 22.04.2020 15:59

Biology, 22.04.2020 15:59






History, 22.04.2020 15:59

Geography, 22.04.2020 15:59






Spanish, 22.04.2020 15:59



English, 22.04.2020 16:00



