Chemistry, 22.04.2020 15:59 Roberto2014
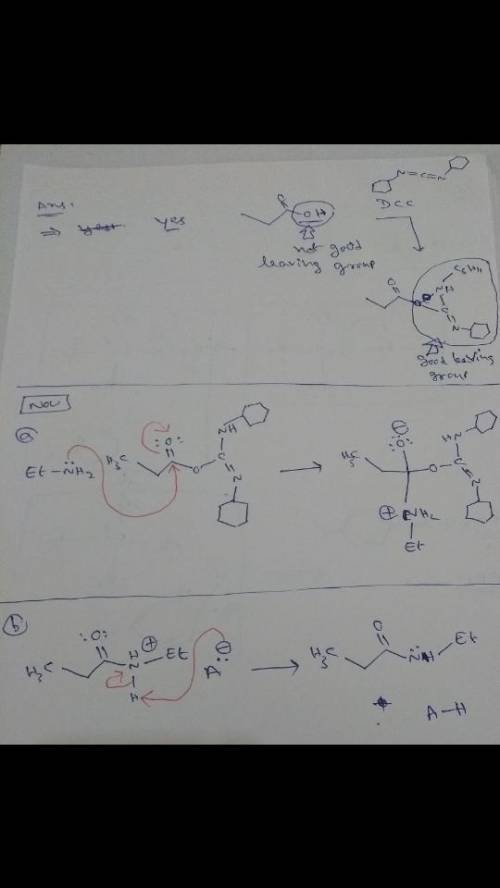
It is difficult to prepare an amide from a carboxylic acid and an amine directly, since an acid-base reaction occurs which renders the amine nitrogen non-nucleophilic. Typically, in such an amide synthesis, the carboxylic acid OH group is first transformed into a better, nonacidic leaving group. In practice, amides are often prepared by treating the carboxylic acid with dicyclohexylcarbodiimide (DCC). The amine is then added and nucleophilic acyl substitution occurs easily because dicyclohexylurea is a good leaving group. This method of amide bond formation is a key step in the laboratory synthesis of peptide bonds (amide bonds) between protected amino acids. Draw curved arrows to show the movement of electrons in this step of the mechanism.

Answers: 3
Another question on Chemistry



Chemistry, 22.06.2019 19:30
Draw the lewis structure for the trisulfur s3 molecule. be sure to include all resonance structures that satisfy the octet rule.
Answers: 3

Chemistry, 22.06.2019 21:30
In science class richard learns that a substance has a boiling point of 230 fahrenheit his teacher ask him to convert this temperature to degrees celsius what is the boiling point of his substance in degrees celsius
Answers: 3
You know the right answer?
It is difficult to prepare an amide from a carboxylic acid and an amine directly, since an acid-base...
Questions



Mathematics, 20.07.2019 17:10


Business, 20.07.2019 17:10

Chemistry, 20.07.2019 17:10

Biology, 20.07.2019 17:10



Social Studies, 20.07.2019 17:10


Physics, 20.07.2019 17:10



Chemistry, 20.07.2019 17:10