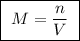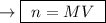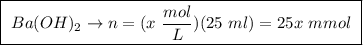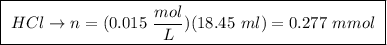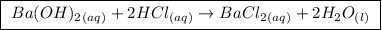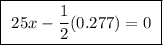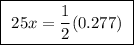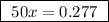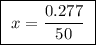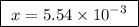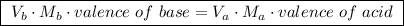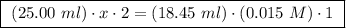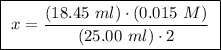Chemistry, 31.07.2019 03:30 kathleensumter5100
A25.00 ml sample of an aqueous solution of ba(oh)2 requires 18.45 ml of 0.01500 m hcl (aq) for its neutralization. what is the molarity of the ba(oh)2 solution?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:30
If a solution is considered basic, then a) the hydroxide ion and hydronium ion concentrations are equal. b) the hydroxide ion concentration is less than the hydronium ion concentration. c) the hydronium ion concentration is greater than the hydroxide ion concentration. d) the hydroxide ion concentration is greater than the hydronium ion concentration.
Answers: 1



Chemistry, 22.06.2019 11:30
If blood contains 150g of hemoglobin per liter of blood, how much hemoglobin would be contained in 10 ml of blood
Answers: 2
You know the right answer?
A25.00 ml sample of an aqueous solution of ba(oh)2 requires 18.45 ml of 0.01500 m hcl (aq) for its n...
Questions


Mathematics, 21.10.2021 17:30


Spanish, 21.10.2021 17:30




History, 21.10.2021 17:50










English, 21.10.2021 18:20

History, 21.10.2021 18:20

Mathematics, 21.10.2021 18:20