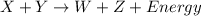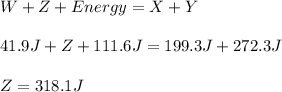Chemistry, 01.07.2019 02:40 osmarirodriguez2079
Reactant x contains 199.3 j of chemical energy. reactant y contains 272.3 j of chemical energy. product w contains 41.9 j of chemical energy. if the reaction loses 111.6 j of chemical energy as it proceeds, how much chemical energy must product z contain? 318.1 j 429.7 j 541.3 j 625.1 j

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:10
Nitric oxide (no) can be formed from nitrogen, hydrogen and oxygen in two steps. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 2 h_2(g) rightarrow 2 nh_3 (g) delta h = -92. kj in the second step, ammonia and oxygen react to form nitric oxide and water: 4 nh_3(g) + 5 o_2(g) rightarrow 4no(g) + 6 h_2o(g) delta h = -905. kj calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 1

Chemistry, 21.06.2019 22:30
Consider the following system at equilibrium. caco3(s) ca2+(aq) + co32–(aq) the addition of which compound will cause a shift in equilibrium because of a common ion effect? ccl4 co2 cuso4 na2co3
Answers: 3


Chemistry, 22.06.2019 08:30
If i initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 k, and then i raise the pressure to 14 atm and increase the temperature to 300 k, what is the new volume of the gas?
Answers: 2
You know the right answer?
Reactant x contains 199.3 j of chemical energy. reactant y contains 272.3 j of chemical energy. prod...
Questions

Mathematics, 29.06.2019 07:30



Computers and Technology, 29.06.2019 07:30

Chemistry, 29.06.2019 07:30


Mathematics, 29.06.2019 07:30

Biology, 29.06.2019 07:30

Health, 29.06.2019 07:30



English, 29.06.2019 07:30

Health, 29.06.2019 07:30

History, 29.06.2019 07:30

Biology, 29.06.2019 07:30


Mathematics, 29.06.2019 07:30

Arts, 29.06.2019 07:30

History, 29.06.2019 07:30

Mathematics, 29.06.2019 07:30