
Chemistry, 13.12.2021 07:10 heybrothwrlogan
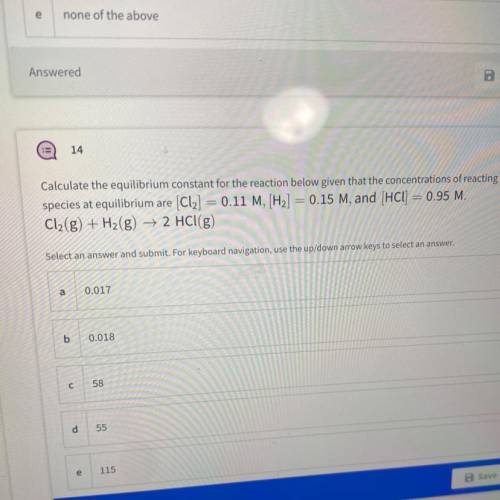
Calculate the equilibrium constant for the reaction below given that the concentrations of reacting
species at equilibrium are (Cl2] = 0.11 M, [H2] = 0.15 M, and (HCl) = 0.95 M.
Cl2(g) + H2(g) → 2 HCl(g)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 11:30
Voltaic cells produce a positive overall charge. what does this indicate? a. the reaction is likely to be endothermic. b. the reaction is spontaneous. c. the reaction is not likely to occur. d. the reaction is not spontaneous.
Answers: 3

Chemistry, 23.06.2019 06:30
Consider the heating curve of h2o and line segments a, b, and c. several changes are taking place at a, b, and c. all but one would be an appropriate description as e move through segments a, b and then c.
Answers: 3

Chemistry, 23.06.2019 08:00
The biosphere of the earth is made up of . a. inorganic b. organic
Answers: 2

You know the right answer?
Calculate the equilibrium constant for the reaction below given that the concentrations of reacting...
Questions



Mathematics, 26.10.2021 23:10

Chemistry, 26.10.2021 23:10

Mathematics, 26.10.2021 23:10

Mathematics, 26.10.2021 23:10










Mathematics, 26.10.2021 23:10

English, 26.10.2021 23:10

Chemistry, 26.10.2021 23:10

Mathematics, 26.10.2021 23:10

Mathematics, 26.10.2021 23:10



