
Chemistry, 07.12.2021 19:40 isaiahcannon5709
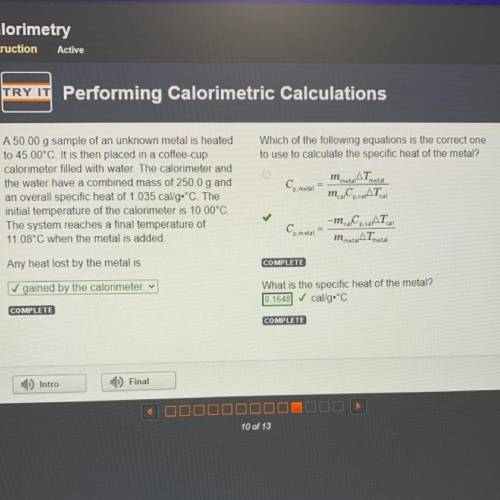
Which of the following equations is the correct one
to use to calculate the specific heat of the metal?
AT
Cp. metal
m. Co. AT
metal
A 50.00 g sample of an unknown metal is heated
to 45.00°C. It is then placed in a coffee-cup
calorimeter filled with water. The calorimeter and
the water have a combined mass of 250.0 g and
an overall specific heat of 1.035 cal/g•°C. The
initial temperature of the calorimeter is 10.00°C.
The system reaches a final temperature of
11.08°C when the metal is added.
metal
cal
Ce metal
-m. C.CAT
m metarATmetal
COMPLETE
Any heat lost by the metal is
✓ gained by the calorimeter v
What is the specific heat of the metal?
cal/g.°C
COMPLETE
DONE

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Read these sentences from the text. near the equator, the tropics receive the most rain on a consistent basis. as a result, the fresh water falling into the ocean decrease the salinity of the surface water in that region. [. .] . . as the salt content of sea water increases, so does its density. what can you infer about how rain affects the density of surface water near the equator?
Answers: 1


Chemistry, 22.06.2019 16:00
How could a student test the effect of removing heat from a gas that is stored in a sealed container? what must occur in order for matter to change states?
Answers: 2

You know the right answer?
Which of the following equations is the correct one
to use to calculate the specific heat of the m...
Questions


Mathematics, 13.04.2021 16:10

Mathematics, 13.04.2021 16:10

Social Studies, 13.04.2021 16:10

Mathematics, 13.04.2021 16:10

French, 13.04.2021 16:10

Computers and Technology, 13.04.2021 16:10


Mathematics, 13.04.2021 16:10


Mathematics, 13.04.2021 16:10

English, 13.04.2021 16:10


Mathematics, 13.04.2021 16:10

History, 13.04.2021 16:10

Chemistry, 13.04.2021 16:10

Mathematics, 13.04.2021 16:10


Social Studies, 13.04.2021 16:10

Biology, 13.04.2021 16:10



