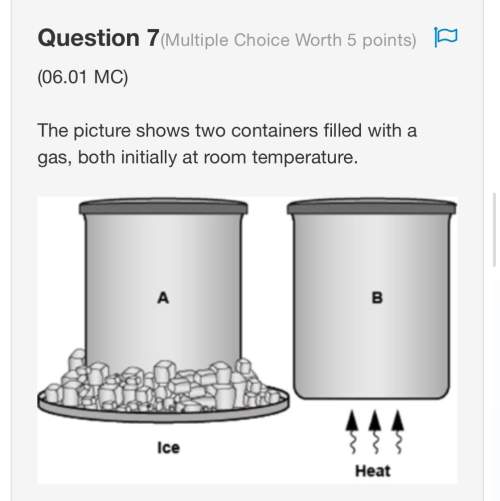
Consider the reaction of
C6H6 + Br2 > C6H5Br + HBr
Balance the reaction above. What is th...

Chemistry, 06.08.2021 18:50 jruiz33106
Consider the reaction of
C6H6 + Br2 > C6H5Br + HBr
Balance the reaction above. What is the limiting reagent if 42.1g of C6H6 react with 73.0 g of Br2

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 11:00
The diagram below shows the different phase transitions that occur in matter. which arrow represents the transition in which dew is formed?
Answers: 1

Chemistry, 23.06.2019 00:30
Balance the following reaction. as2s3 + 9o2 → 2as2o3 + so2
Answers: 2

Chemistry, 23.06.2019 02:20
Which of the following will cause an increase in the acceleration of an object? increase force decrease force increase mass decrease mass
Answers: 1

Chemistry, 23.06.2019 07:30
Which statement is actually true about the relationship between activation energy and reaction rates? low activation energy barriers result in low rates. high activation energy barriers result in low rates. low activation energy barriers result in no reaction. high activation energy barriers result in no reaction.
Answers: 2
You know the right answer?
Questions

Mathematics, 16.07.2019 06:10

Mathematics, 16.07.2019 06:10

English, 16.07.2019 06:10












Computers and Technology, 16.07.2019 06:10