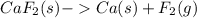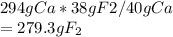Chemistry, 07.07.2021 01:30 Dkhaurithompson
A sample of calcium fluoride was decomposed into the constituent elements. Write a balanced chemical equation for the decomposition reaction. If the sample produced 294 mg of calcium, how many g of fluorine were formed

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:30
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen, and 54.2 grams of oxygen. in an experiment, the molar mass of the compound was determined to be 118.084 g/mol. what is the molecular formula of the compound? for both questions, show your work or explain how you determined the formulas by giving specific values used in calculations.
Answers: 3

Chemistry, 22.06.2019 08:00
Me i dont know what to do! the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 12:00
Which of the following units is not an official si unit? mole liter kilogram ampere
Answers: 1

Chemistry, 22.06.2019 20:30
A40 kilogram skier starts at the top of a 12 meter high slope. at the bottom, she is travelling 10 meters per second. how much energy does she lose to friction
Answers: 2
You know the right answer?
A sample of calcium fluoride was decomposed into the constituent elements. Write a balanced chemical...
Questions

English, 14.08.2021 15:10

Mathematics, 14.08.2021 15:10


Mathematics, 14.08.2021 15:10

Social Studies, 14.08.2021 15:10

Mathematics, 14.08.2021 15:10

Computers and Technology, 14.08.2021 15:10

Computers and Technology, 14.08.2021 15:10


English, 14.08.2021 15:10


Mathematics, 14.08.2021 15:10



Mathematics, 14.08.2021 15:10