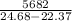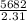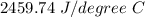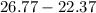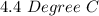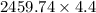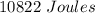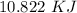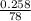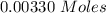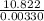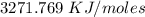Chemistry, 16.06.2021 23:10 ejcastilllo
Two experiments were conducted in a bomb calorimeter. The first one to determine the heat capacity of the calorimeter, the second the heat of combustion of the carcinogenic substance benzene (C6H6). a. In the first experiment, the temperature rises from 22.37 o C to 24.68 o C when the calorimeter absorbs 5682 J of heat. Determine the heat capacity of the calorimeter. Page 3 of 4 b. In the second experiment, the combustion of 0.258 g of benzene increases the temperature from 22.37 o C to 26.77 o C. Determine the heat of combustion for 1 mol of benzene.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Match the following items. 1. high-intensity bundle of energy being emitted from some decaying nuclei gamma ray 2. particle radiating from the nucleus of some atoms beta particle 3. negative particle identical to an electron but radiating from a decaying nucleus alpha particle
Answers: 1

Chemistry, 22.06.2019 19:30
Use the periodic table to find the molar mass of each element. molar mass h = g/mol molar mass s = g/mol molar mass o = g/mol
Answers: 3

Chemistry, 22.06.2019 22:30
What must be in balance for temperatures to remain constant?
Answers: 1

Chemistry, 23.06.2019 01:00
Which of the following is a physical change? a.burning a piece of wood b.sawing a piece of wood in half c.rust forming on an iron fence d.a copper roof changing color from orange to green
Answers: 1
You know the right answer?
Two experiments were conducted in a bomb calorimeter. The first one to determine the heat capacity o...
Questions






Geography, 28.09.2019 17:30





Biology, 28.09.2019 17:30



Mathematics, 28.09.2019 17:30


Mathematics, 28.09.2019 17:50


Mathematics, 28.09.2019 17:50

Biology, 28.09.2019 17:50