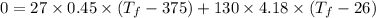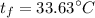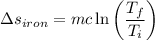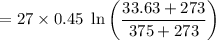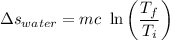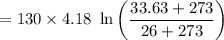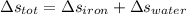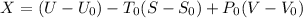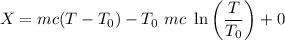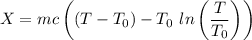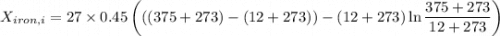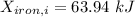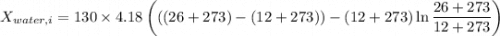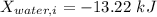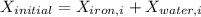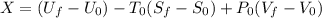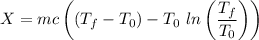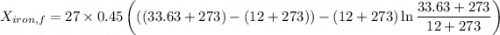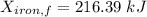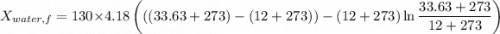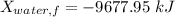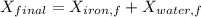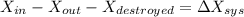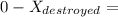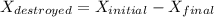A 27 kg iron block initially at 375 C is quenched in an insulated tank that contains 130kg of water at 26 C. Assume the water that vaporizes during the process condenses back in the tank and the surroundings are at 12 C and 125 kPa. The specific heats of iron and water are 0.45 kJ/kgK and 4.18 kJ/kgK, respectively. a: Determine the final equilibrium temperature. b: Determine the entropy change of the combined system at the initial state. c: Determine the exergy of the combined system at the initial state. d: Determine the waste work potential during this process.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:00
Which solution is an example of a nonelectrolyte? a. iodine in hexane b. sodium nitrate in waterc. acetic acid in waterd. hydrogen chloride in water
Answers: 2


Chemistry, 22.06.2019 15:20
Identify arrows pointing to bonding electrons. done h-0-0-h ) intro
Answers: 3

Chemistry, 22.06.2019 16:00
If 15 drops of ethanol from a medical dropper weight 0.60g, how many drops does it takes from a dropper to dispense 1.0ml of ethanol? the density of ethanol is 0.80g/ml
Answers: 1
You know the right answer?
A 27 kg iron block initially at 375 C is quenched in an insulated tank that contains 130kg of water...
Questions


English, 22.10.2020 08:01





Mathematics, 22.10.2020 08:01


Physics, 22.10.2020 08:01


Biology, 22.10.2020 08:01


Health, 22.10.2020 08:01


Health, 22.10.2020 08:01



Mathematics, 22.10.2020 08:01

History, 22.10.2020 08:01

Geography, 22.10.2020 08:01

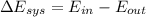
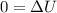
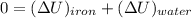
![$0=[mc(T_f-T_i)_{iron}] + [mc(T_f-T_i)_{water}]$](/tpl/images/1350/3800/c1f46.png)