
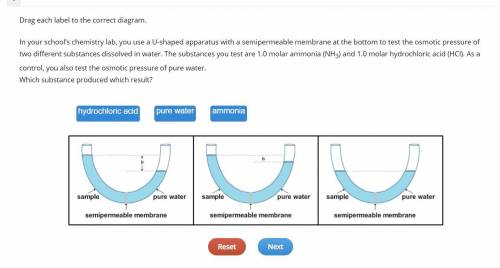
Drag each label to the correct diagram.
In your school’s chemistry lab, you use a U-shaped apparatus with a semipermeable membrane at the bottom to test the osmotic pressure of two different substances dissolved in water. The substances you test are 1.0 molar ammonia (NH3) and 1.0 molar hydrochloric acid (HCl). As a control, you also test the osmotic pressure of pure water.
Which substance produced which result?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 09:30
The chart shows the bid provided by four contractors to complete a job. which contractor is the most cost-effective?
Answers: 3


You know the right answer?
Drag each label to the correct diagram.
In your school’s chemistry lab, you use a U-shaped apparatu...
Questions

English, 18.01.2021 05:30

Physics, 18.01.2021 05:30

Mathematics, 18.01.2021 05:30


History, 18.01.2021 05:30

Mathematics, 18.01.2021 05:30

Arts, 18.01.2021 05:30

Mathematics, 18.01.2021 05:30






English, 18.01.2021 05:30




Chemistry, 18.01.2021 05:30

Social Studies, 18.01.2021 05:30

Geography, 18.01.2021 05:30



