Someone please help!!
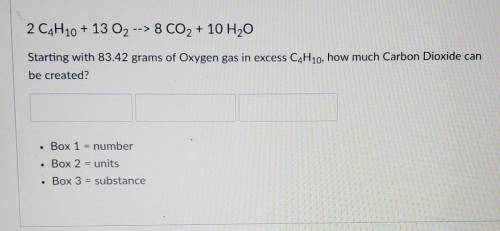
2 C4H10 + 13 O2 --> 8 CO2 + 10 H2O
Starting with 83.42 grams of Oxyg...

Chemistry, 07.05.2021 22:00 xMABRYx1991
Someone please help!!
2 C4H10 + 13 O2 --> 8 CO2 + 10 H2O
Starting with 83.42 grams of Oxygen gas in excess C4H10, how much Carbon Dioxide can be created?
Box 1 = number
Box 2 = units
Box 3 = substance

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 23:00
Will mark brainliest26. which of these statements are true? (3 points)a. gases are compressibleb. gases fill their containers completelyc. the pressure of a gas is independent of the temperatured. gases have masse. gases exert pressuref. the pressure of a gas is dependent on the volumeg. gas pressure results from the collisions between gas particlesh. gases have a definite volume and shape
Answers: 1

Chemistry, 22.06.2019 17:40
If 3 moles of a compound use 24 j of energy in a reaction, what is the a hreaction in j/mol?
Answers: 1

Chemistry, 22.06.2019 20:10
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3

Chemistry, 23.06.2019 00:00
What is the approximate mass of 25 cm3 of silver, if the density is 10.5 g/cm3? a. 0.42 g b. 2.4 g c. 42 g d. 260 g
Answers: 1
You know the right answer?
Questions

Business, 07.11.2020 03:40


Biology, 07.11.2020 03:40

Social Studies, 07.11.2020 03:40

Mathematics, 07.11.2020 03:40

History, 07.11.2020 03:40

Mathematics, 07.11.2020 03:40



Arts, 07.11.2020 03:40

Health, 07.11.2020 03:40

Mathematics, 07.11.2020 03:40



Biology, 07.11.2020 03:40


Geography, 07.11.2020 03:40

Social Studies, 07.11.2020 03:40

SAT, 07.11.2020 03:40



