Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3

Chemistry, 22.06.2019 19:00
Which statement best describes what happens when molecular compounds melt
Answers: 1

Chemistry, 22.06.2019 20:00
What is the molar mass of the anhydrous compound? answer using four significant figures. 36.02 g/mol 120.15 g/mol 156.12 g/mol
Answers: 1

You know the right answer?
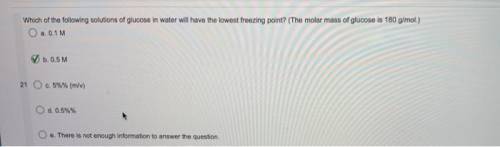
Which of the following solutions of glucose in water will have the lowest freezing point?
(The mola...
Questions

Biology, 14.01.2020 19:31


Mathematics, 14.01.2020 19:31

Mathematics, 14.01.2020 19:31

Mathematics, 14.01.2020 19:31

Computers and Technology, 14.01.2020 19:31


English, 14.01.2020 19:31

Engineering, 14.01.2020 19:31

Mathematics, 14.01.2020 19:31

History, 14.01.2020 19:31




Arts, 14.01.2020 19:31


Biology, 14.01.2020 19:31