
Chemistry, 03.05.2021 19:00 kcutler8603
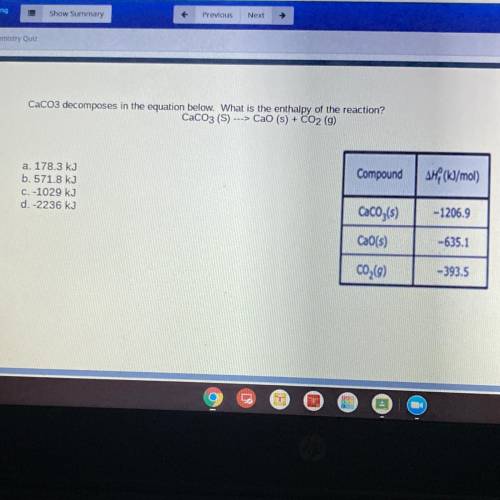
CaCO3 decomposes in the equation below. What is the enthalpy of the reaction?
CaCO3 (S) ---> Cao (s) + CO2 (g)
Compound
AH (kJ/mol)
a. 178.3 kJ
b. 571.8 kJ
C. -1029 kJ
d.-2236 kJ
CaCO3(s)
-1206.9
CaO(s)
-635.1
C02(9)
-393.5

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 08:40
What is the value of keq for the reaction expressed in scientific notation?
Answers: 1

Chemistry, 22.06.2019 12:30
The melting point of sulfur is 115 °c and its boiling point is 445 °c. what state would sulfur be in at 200 °c?
Answers: 1

Chemistry, 22.06.2019 14:00
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 1

Chemistry, 22.06.2019 19:20
The equation picture below shows which type of nuclear reaction u 235 + n x e 134 + sr 100 + 2n
Answers: 1
You know the right answer?
CaCO3 decomposes in the equation below. What is the enthalpy of the reaction?
CaCO3 (S) ---> Ca...
Questions


Geography, 07.12.2020 22:20





Mathematics, 07.12.2020 22:20




Mathematics, 07.12.2020 22:20

Mathematics, 07.12.2020 22:20

Law, 07.12.2020 22:20



Chemistry, 07.12.2020 22:20

Mathematics, 07.12.2020 22:20



English, 07.12.2020 22:20



