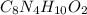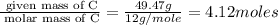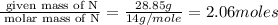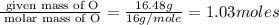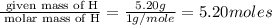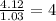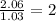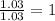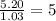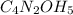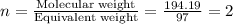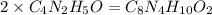Chemistry, 19.04.2021 15:40 impura12713
Caffeine is a compound found in some natural coffees and teas and in some colas. a. Determine the empirical formula for caffeine, using the following composition of a 100.00-g sample. 49.47 grams of carbon, 28.85 grams of nitrogen, 16.48 grams of oxygen, and 5.20 grams of hydrogen b. If the molar mass of caffeine is 194.19 g/mol, calculate its molecular formula.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Omg imgonnafailnfiedkla use complete sentences to explain how the mass of hydrogen is conserved during cellular respiration.
Answers: 1


Chemistry, 22.06.2019 08:30
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2

Chemistry, 22.06.2019 13:30
The atomic number, or number, is the described as the number of in the nucleus of an chemical element.
Answers: 1
You know the right answer?
Caffeine is a compound found in some natural coffees and teas and in some colas. a. Determine the em...
Questions





Chemistry, 05.05.2020 20:19




History, 05.05.2020 20:19

Mathematics, 05.05.2020 20:19


Law, 05.05.2020 20:19



Mathematics, 05.05.2020 20:19





Mathematics, 05.05.2020 20:19