In the following molecules, the primary intermolecular attractive force is
...

Chemistry, 05.04.2021 22:50 deedivinya
In the following molecules, the primary intermolecular attractive force is
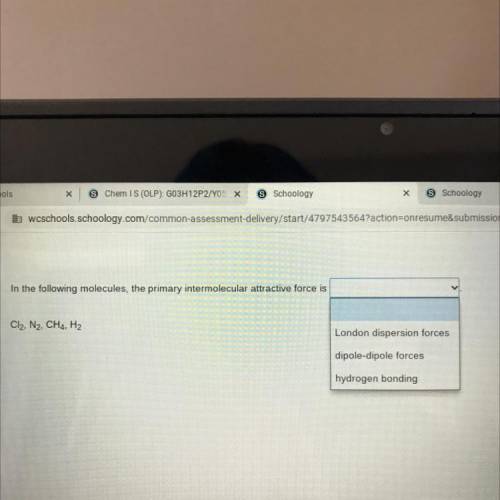

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 17:00
Look at the reaction below: ca(hco3)2 --> caco3 + co2 + h2o first, balance the reaction. once balanced, use dimensional analysis or another method to find out how many moles of carbon dioxide will be produced if we start with 16.5 moles of calcium bicarbonate (calcium hydrogen carbonate). = mol of co2 number needs to be reported to three significant figures.
Answers: 1


Chemistry, 22.06.2019 17:00
Astable electron arrangement for an atom is one that does not easily change. how is this arrangement arrived at? a. valence electrons are transferred or shared to create a full outer shell of electrons. b. valence electrons are discarded into space to create a full outer shell of electrons. c. protons (positive charge) pair with valence electrons (negative charge) to create a strong bond. d. outer shells with valence electrons are transferred or shared.
Answers: 2

Chemistry, 23.06.2019 03:30
If 2 molecules of one reactant combine with 3 molecules of another to produce 5 molecules of a product, then what is the representation of the reaction?
Answers: 1
You know the right answer?
Questions


Biology, 16.12.2020 01:00


Chemistry, 16.12.2020 01:00


Biology, 16.12.2020 01:00

Biology, 16.12.2020 01:00

Mathematics, 16.12.2020 01:00

Social Studies, 16.12.2020 01:00

Mathematics, 16.12.2020 01:00


Mathematics, 16.12.2020 01:00



Spanish, 16.12.2020 01:00

Mathematics, 16.12.2020 01:00


History, 16.12.2020 01:00




