What is the mole ratio of all the substances represented in this figure? AKS 4f
...

Chemistry, 09.03.2021 14:00 19thomasar
What is the mole ratio of all the substances represented in this figure? AKS 4f
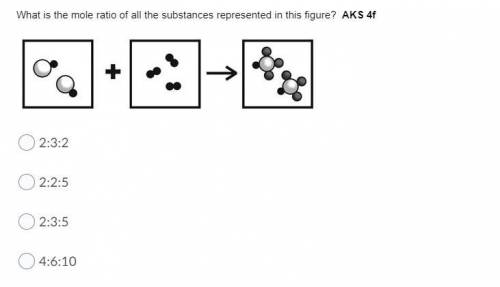

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 18:00
Answer asap need it by wednesday morning carry out the following calculations on ph and ka of from data. i. calculate the ph of 0.02m hcl ii. calculate the ph of 0.036m naoh iii. calculate the ph of 0.36m ca(oh)2 iv. calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 v. calculate ka for weak acid ha which has a ph of 3.65 at 0.30m concentration vi. calculate the ka of a solution made by mixing 15.0 cm3 0.2m ha and 60.0 cm3 0.31m a-. [ph= 3.80] vii. calculate the ph of a solution made by mixing 15.0 cm3 0.1m naoh and 35.0 cm3 0.2m hcooh. [ka = 1.82 x 10-4 m]
Answers: 1

Chemistry, 23.06.2019 00:00
2-bromo-2-methylbutane undergoes an e1 elimination reaction in the presence of ethanol. in the next reaction only one of the possible products is represented. although the product shown is not the major product of the reaction, notice that there is more than one way it can be produced. complete the mechanism and draw the missing substances.
Answers: 1

Chemistry, 23.06.2019 09:00
What sources of error may have contributed to the percent yield not being 100 percent? think about things that may have led to inaccurate measurements or where mass of the product could have been lost if this experiment was conducted in a physical laboratory.
Answers: 2

Chemistry, 23.06.2019 09:20
Asolution of naoh has a concentration of 25.00% by mass. what mass of naoh is present in 0.250 g of this solution? use the periodic table in the toolbar if needed. 0.0625 g what mass of naoh must be added to the solution to increase the concentration to 30.00% by mass? g
Answers: 2
You know the right answer?
Questions

Mathematics, 05.11.2020 04:30

Mathematics, 05.11.2020 04:30

History, 05.11.2020 04:30

English, 05.11.2020 04:30

Mathematics, 05.11.2020 04:30


Physics, 05.11.2020 04:30

Social Studies, 05.11.2020 04:30

Physics, 05.11.2020 04:30

Mathematics, 05.11.2020 04:30




Chemistry, 05.11.2020 04:30


English, 05.11.2020 04:30



Physics, 05.11.2020 04:30

Arts, 05.11.2020 04:30