
Chemistry, 04.03.2021 21:40 kerstynsharp08
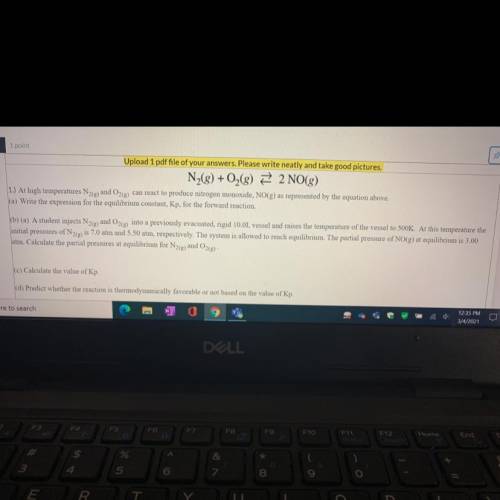
N2 + O2 —> 2NO
b) A student injects N2(g)and O2(g) into a previously evacuated, rigid 10.0L vessel and raises the temperature of the vessel to 500K. At this temperature the
initial pressures of N2(g) is 7.0 atm and 5.50 atm, respectively. The system is allowed to reach equilibrium. The partial pressure of NO(g) at equilibrium is 3.00
atm. Calculate the partial pressures at equilibrium for N2(e) and O2(g)

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 19:30
Water molecules have a strong attraction to each other because of hydrogen bonding, allowing water to move against gravity up a plant's stem through capillary action. true false
Answers: 2


Chemistry, 22.06.2019 00:00
Acurium-245 nucleus is hit with a neutron and changes as shown by the equation. complete the equation by filling in the missing parts. 52
Answers: 2

Chemistry, 23.06.2019 01:00
Wind and moving water provide energy. chemical mechanical thermal none of the above
Answers: 1
You know the right answer?
N2 + O2 —> 2NO
b) A student injects N2(g)and O2(g) into a previously evacuated, rigid 10.0L vess...
Questions

Mathematics, 04.02.2020 12:53


Biology, 04.02.2020 12:53


Arts, 04.02.2020 12:53


Mathematics, 04.02.2020 12:53

Geography, 04.02.2020 12:53





English, 04.02.2020 12:53

Social Studies, 04.02.2020 12:53


Mathematics, 04.02.2020 12:53

Health, 04.02.2020 12:53


English, 04.02.2020 12:53



