Please help!!
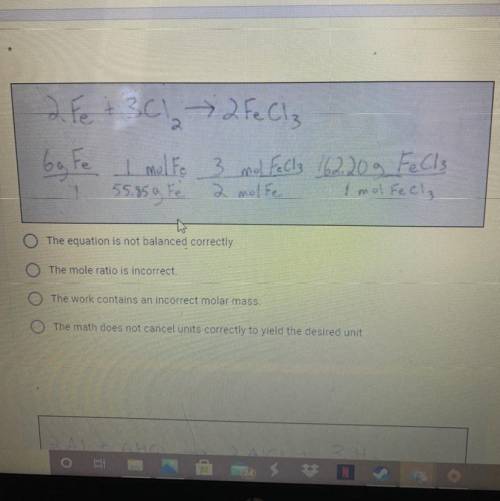
4
4 NH₃ +
+502 + 3 NO + 6H₂O
4 mol NH3
5 mol O2
4 mol N...

Chemistry, 03.03.2021 21:30 graymonky12
Please help!!
4
4 NH₃ +
+502 + 3 NO + 6H₂O
4 mol NH3
5 mol O2
4 mol NH3
1
O The equation is not balanced correctly
The mole ratio is incorrect.
The work contains an incorrect molar mass.
The math does not cancel units correctly to yield the desired unit

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 12:30
The melting point of sulfur is 115 °c and its boiling point is 445 °c. what state would sulfur be in at 200 °c?
Answers: 1

Chemistry, 23.06.2019 03:40
The following questions a24 - a26 relate to 100 ml of 0.0150 m solution of benzoic acid (c6h3cooh). ka(c6h3cooh) = 6.4 x 10^-5. what is the ph of the solution after the addition of 1 x 10^-3 moles of naoh? you may assume no volume change to the solution upon addition of the naoh.
Answers: 2

Chemistry, 23.06.2019 08:30
Explain how to convert from one unit to another in the metric system.
Answers: 3

Chemistry, 23.06.2019 08:40
Which statement is true according to the kinetic theory? a. molecules of different gases with the same mass and temperature always have the same average density. b. molecules of different gases with the same mass and temperature always have the same average volume. c. molecules of different gases with the same mass and temperature always have the same pressure. d. molecules of different gases with the same mass and temperature always have the same molecular mass. e. molecules of different gases with the same mass and temperature always have the same average kinetic energy.
Answers: 1
You know the right answer?
Questions


Chemistry, 13.10.2021 01:30

Mathematics, 13.10.2021 01:30





Mathematics, 13.10.2021 01:30

English, 13.10.2021 01:30


Health, 13.10.2021 01:30

Physics, 13.10.2021 01:30


Biology, 13.10.2021 01:30

Mathematics, 13.10.2021 01:30


Social Studies, 13.10.2021 01:30





