
Chemistry, 09.02.2021 01:50 JamesLachoneus
Liquid octane CH3CH26CH3 will react with gaseous oxygen O2 to produce gaseous carbon dioxide CO2 and gaseous water H2O. Suppose 2.3 g of octane is mixed with 12.4 g of oxygen. Calculate the minimum mass of octane that could be left over by the chemical reaction. Round your answer to 2 significant digits.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:50
Which statement is a reason to support population regulation? a) it is unethical for us to control birth control rates b) humans have the freedom to produce as many children as desired c) the gap between the rich and poor has been narrowing since 1960 d) billions more people on the earth will intensify many environmental and social problems
Answers: 1

Chemistry, 22.06.2019 10:50
A100 kmol/h stream that is 97 mole% carbon tetrachloride (ccl4) and 3% carbon disulfide (cs2) is to be recovered from the bottom of a distillation column. the feed to the column is 16 mole% cs2 and 84% ccl4, and 2% of the ccl4 entering the column is contained in the overhead stream leaving the top of the column. calculate the mass and mole fractions of ccl4 in the overhead stream, and determine the molar flow rates of ccl4 and cs2 in the overhead and feed streams. 12. mw_ccla- 153.82; mw_cs2-76.14.
Answers: 3


You know the right answer?
Liquid octane CH3CH26CH3 will react with gaseous oxygen O2 to produce gaseous carbon dioxide CO2 and...
Questions




Mathematics, 02.09.2021 19:20




Mathematics, 02.09.2021 19:20

Mathematics, 02.09.2021 19:20




English, 02.09.2021 19:20

Mathematics, 02.09.2021 19:20


Computers and Technology, 02.09.2021 19:20




Mathematics, 02.09.2021 19:20

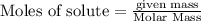
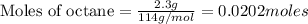
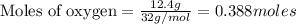
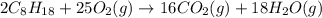
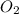
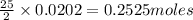 of
of 

