
Chemistry, 08.02.2021 21:40 sillyvanna
PLEASE HELP ASAP WILL GIVE BRAINLIEST!!
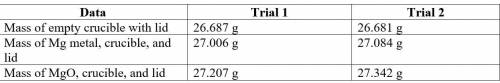
2. Subtract the mass of the crucible and lid (row 1 in the chart) from the total mass of Mg, crucible, and lid (row 2 in the chart) to find the mass of magnesium for each trial.
• Trial 1:
• Trial 2:
3. Subtract the mass of the crucible and lid (row 1 in the chart) from the total mass of MgO, crucible, and lid (row 3 in the chart) to find the mass of magnesium oxide for each trial. This is the actual yield of magnesium oxide for each trial.
• Trial 1:
• Trial 2:
4. Magnesium is the limiting reactant in this experiment. Calculate the theoretical yield of MgO for each trial.
• Trial 1:
• Trial 2:
5. Determine the percent yield of MgO for your experiment for each trial.
• Trial 1:
• Trial 2:
6. Determine the average percent yield of MgO for the two trials.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 08:30
Which of the following would have less momentum than a 52 kg cheetah running at 10 m/s?
Answers: 2

Chemistry, 22.06.2019 09:00
Chen drew a diagram to compare the ways in which different organisms obtain nitrogen. which label belongs to the area marked z?
Answers: 3

Chemistry, 22.06.2019 09:10
Select the correct answer from each drop-down menu.describe what happens to a carbon-11 atom when it undergoes positron emission.the decay of a carbon-11 atom _1_, and this causes it to emit _2_.options for 1: > changes a neutron into a proton> changes a proton into a neutron> is hit with a neutron> reconfigures its protons and neutronsoptions for 2: > a negatively charged electron-sized particle> a positively charged election-sized particle> two atoms and several neutrons> two neutrons and two protons
Answers: 3

Chemistry, 22.06.2019 16:30
How many moles of sulfuric acid (h2so4) are needed to react completely with 6.8 moles of lithium hydroxide (lioh)? 2lioh + h2so4 → li2so4 + 2h2o a. 3.4 mol h2so4b. 6.8 mol h2so4 c. 10.2 mol h2so4 d. 13.6 mol h2so4
Answers: 3
You know the right answer?
PLEASE HELP ASAP WILL GIVE BRAINLIEST!!
2. Subtract the mass of the crucible and lid (row 1 in the...
Questions

Mathematics, 04.08.2019 19:10

Biology, 04.08.2019 19:10

Computers and Technology, 04.08.2019 19:10


English, 04.08.2019 19:10


Spanish, 04.08.2019 19:10

Mathematics, 04.08.2019 19:10


History, 04.08.2019 19:10

Mathematics, 04.08.2019 19:10

Social Studies, 04.08.2019 19:10

Business, 04.08.2019 19:10

Physics, 04.08.2019 19:20

Chemistry, 04.08.2019 19:20




Social Studies, 04.08.2019 19:20

Mathematics, 04.08.2019 19:20



