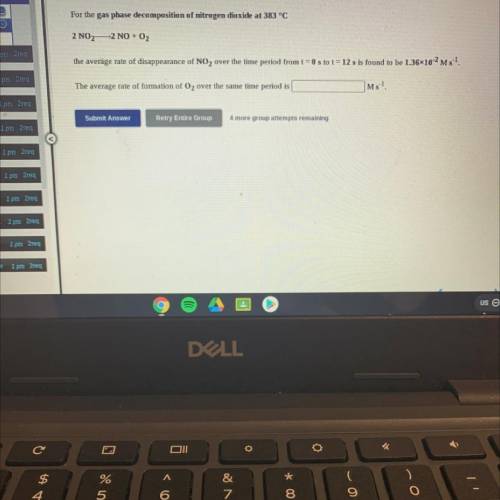
For the gas phase decomposition of nitrogen dioxide at 383 °C
2 NO2—2 NO + O2
the average rat...

Chemistry, 02.02.2021 03:20 tyneshiajones124
For the gas phase decomposition of nitrogen dioxide at 383 °C
2 NO2—2 NO + O2
the average rate of disappearance of NO2 over the time period from t = 0 s to t = 12 s is found to be 1.3610-2 Ms 1.
The average rate of formation of O2 over the same time period is
Ms-1

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:50
Significant figures are digits read directly from the measuring instrument plus one more digit, which is __ by the observer.
Answers: 2

Chemistry, 22.06.2019 12:30
The melting point of sulfur is 115 °c and its boiling point is 445 °c. what state would sulfur be in at 200 °c?
Answers: 1

Chemistry, 22.06.2019 23:30
With the largest atoms and the smallest number of valence electrons and with the smallest atoms and the greatest number of valence electrons are the most reactive. a. nonmetals; metals b. nonmetals; transition elements c. transition elements; metals d. metals; nonmetals
Answers: 3

Chemistry, 23.06.2019 00:00
Total the mass on the syringe. record it in the correct row of the data table. kg done click and drag weights to change the pressure. click the syringe to zoom in and see the volume. intro
Answers: 3
You know the right answer?
Questions



Biology, 02.09.2020 04:01








Mathematics, 02.09.2020 04:01





Mathematics, 02.09.2020 04:01

Mathematics, 02.09.2020 04:01

Biology, 02.09.2020 04:01




