
Chemistry, 29.01.2021 19:40 adamgala3885
How many moles of H2 are needed to produce 18 miles NH3 in the reaction below ? N2+ 3H2 ——-> 2 NH3
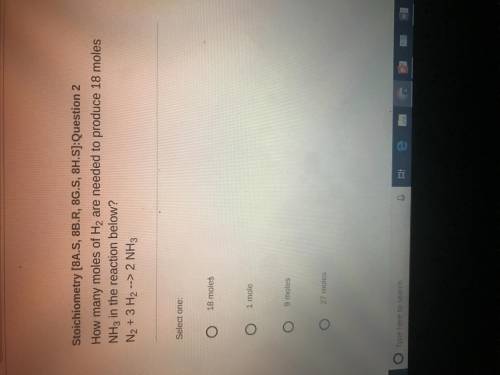

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 20:30
Consider the following unbalanced equation for the combustion of hexane: αc6h14(g)+βo2(g)→γco2(g)+δh2o(g) part a balance the equation. give your answer as an ordered set of numbers α, β, γ, use the least possible integers for the coefficients. α α , β, γ, δ = nothing request answer part b determine how many moles of o2 are required to react completely with 5.6 moles c6h14. express your answer using two significant figures. n n = nothing mol request answer provide feedback
Answers: 2

Chemistry, 22.06.2019 22:30
Draw the aromatic compound toluene (methylbenzene). show all hydrogen atoms, including those on the ring.
Answers: 1

Chemistry, 22.06.2019 22:30
Amedication is given at a dosage of 3.000 mg of medication per kg of body weight. if 0.1500 g of medication is given, then what was the patient's weight in pounds (lbs)? there are 453.59g in 1 lb.
Answers: 2

Chemistry, 22.06.2019 22:30
Why is it possible for different microorganisms to extract energy not only from carbohydrates and other biological molecules but from a large variety of substances?
Answers: 1
You know the right answer?
How many moles of H2 are needed to produce 18 miles NH3 in the reaction below ?
N2+ 3H2 ——-> 2 N...
Questions









Arts, 22.06.2019 00:30

World Languages, 22.06.2019 00:30

Business, 22.06.2019 00:30



History, 22.06.2019 00:30

Social Studies, 22.06.2019 00:30

Mathematics, 22.06.2019 00:30



English, 22.06.2019 00:30

Mathematics, 22.06.2019 00:30



