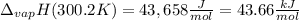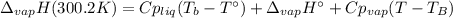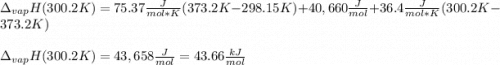Chemistry, 25.01.2021 20:40 PastelHibiscus
The heat of vaporization of water at the normal boiling point, 373.2 K, is 40.66 kJ/mol. The molar heat capacity of liquid water is 75.37 J K-1 mol-1 and that of gaseous water is 36.4 J K-1 mol-1. Assume that these values are independent of temperature. What is the heat of vaporization of water at 300.2 K?

Answers: 3
Another question on Chemistry




Chemistry, 22.06.2019 06:00
How much would the freezing point of water decrease if 4 mol of sugar were added to 1 kg of water(k=1.86 c/mol/kg for water and i=1 for sugar
Answers: 1
You know the right answer?
The heat of vaporization of water at the normal boiling point, 373.2 K, is 40.66 kJ/mol. The molar h...
Questions




Biology, 24.07.2020 03:01





Chemistry, 24.07.2020 03:01

Geography, 24.07.2020 03:01

Business, 24.07.2020 03:01

Mathematics, 24.07.2020 03:01


Mathematics, 24.07.2020 03:01





History, 24.07.2020 03:01