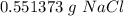1 Na2CO3(aq) + 1 CaCl2(aq) → 1 CaCO3(s) + 2 NaCl(aq) 4. Use the balanced chemical equation from the last question to solve this situation: You combine 0.5 grams of Na2CO3 with excess CaCl2. How many grams of NaCl would you expect this reaction to produce? Show all work below.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:30
What three things determine the shape and size of a puddle when water is poured out onto a surface
Answers: 2

Chemistry, 22.06.2019 12:30
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1

Chemistry, 22.06.2019 23:30
The density of benzene at 15 °c is 0.8787 g/ml. calculate the mass of 0.1500 l of benzene at this temperature. enter your answer in terms of grams
Answers: 2

You know the right answer?
1 Na2CO3(aq) + 1 CaCl2(aq) → 1 CaCO3(s) + 2 NaCl(aq) 4. Use the balanced chemical equation from the...
Questions

Biology, 22.09.2019 04:30


Chemistry, 22.09.2019 04:30

Mathematics, 22.09.2019 04:30


Mathematics, 22.09.2019 04:30

Spanish, 22.09.2019 04:30


SAT, 22.09.2019 04:30

Mathematics, 22.09.2019 04:30


Social Studies, 22.09.2019 04:30

Physics, 22.09.2019 04:30

Computers and Technology, 22.09.2019 04:30


Mathematics, 22.09.2019 04:30


Mathematics, 22.09.2019 04:30


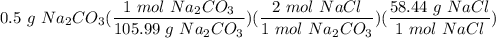 Multiply/Divide:
Multiply/Divide: