Answers: 3
Another question on Chemistry



Chemistry, 23.06.2019 00:00
Before it was launched, a helium-filled balloon had a pressure of 201 kpa at a temperature of 27°c. at an altitude of 15,000 m, the pressure had decreased to 2.5 kpa and the temperature had dropped to -14 °c. the volume of the balloon increased to 59.3 m3. what is the original volume of the balloon? 13 m3 0.85 m3 0.077 m3 1.17 m3
Answers: 3

Chemistry, 23.06.2019 00:30
Titration reveals that 11.6 ml of 3.0m sulfuric acid are required to neutralize the sodium hydroxide in 25.00ml of naoh solution. what is the molarity of the naoh solution?
Answers: 1
You know the right answer?
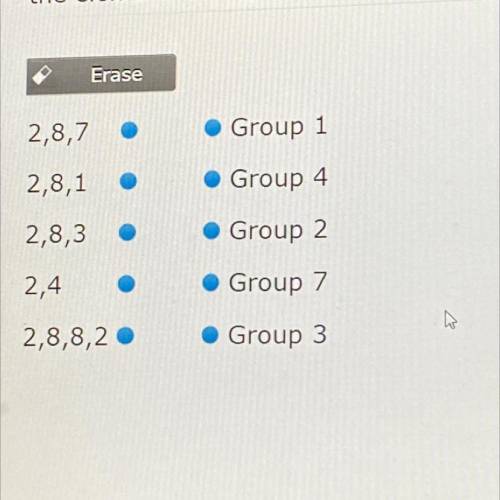
Connect the group to the configuration. pleas help !!
...
...
Questions

Physics, 06.11.2020 08:20

Business, 06.11.2020 08:20

Mathematics, 06.11.2020 08:20




Mathematics, 06.11.2020 08:20



Mathematics, 06.11.2020 08:20

History, 06.11.2020 08:20

English, 06.11.2020 08:20


History, 06.11.2020 08:20

Mathematics, 06.11.2020 08:20


Biology, 06.11.2020 08:20

Arts, 06.11.2020 08:20