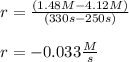Chemistry, 11.01.2021 15:50 asmith219771
Calculate the average rate of decomposition of NH4NO2 by the reaction 2NH4O5(aq) --> N2(g) H2O(g) at the following time interval. At time 250 s, the concentration of NH4NO2 is 4.12 M. At time 330 s, the concentration of NH4NO2 is 1.48 M

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:10
Starch and are common polysaccharide carbohydrates found in plants. sucrose glycogen fructose cellulose
Answers: 3

Chemistry, 21.06.2019 22:30
1.aluminum chloride (alcl3), and sodium hydroxide (naoh) can react to form aluminum hydroxide (al(oh)3) and sodium chloride (nacl). you have 13.4 g of aluminum chloride and 10.0 g of sodium hydroxide. answer the following questions: •what is the balanced equation for this reaction? •if you use all 13.4 g of aluminum chloride, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •if you use all 10.0 g of sodium hydroxide, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •how many grams of aluminum hydroxide will actually be made? which reagent is limiting? explain your answer.
Answers: 1


Chemistry, 23.06.2019 02:00
The bohr model of the atom explained why emission spectra are discrete. it could also be used to explain the photoelectric effect. which is a correct explanation of the photoelectric effect according to the model?
Answers: 3
You know the right answer?
Calculate the average rate of decomposition of NH4NO2 by the reaction 2NH4O5(aq) --> N2(g) H2O(g)...
Questions

English, 11.07.2019 11:00

Chemistry, 11.07.2019 11:00

English, 11.07.2019 11:00

Advanced Placement (AP), 11.07.2019 11:00

Mathematics, 11.07.2019 11:00

Social Studies, 11.07.2019 11:00


Mathematics, 11.07.2019 11:00


Biology, 11.07.2019 11:00



Mathematics, 11.07.2019 11:00




English, 11.07.2019 11:00



Mathematics, 11.07.2019 11:00

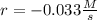
![r=\frac{\Delta [NH_4NO_2 ]}{\Delta t}](/tpl/images/1025/9856/b44bf.png)