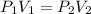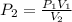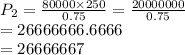The pressure inside of a sealed syringe of hydrogen gas is 80.0KPa with a volume of 250 Liters. What is the pressure inside
of the syringe after the plunger is pulled back far enough to make final volume of the syringe 0.750 Liters? (assume the
temperature and amount of gas in the container are held constant)

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:30
Which element is the least metallic between cadmium, silver, zinc, or iron?
Answers: 1

Chemistry, 22.06.2019 10:20
Gwhich r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? which r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? −ch2−oh −ch2−o||c−nh2 −ch2−coo− −ch2−ch2−ch2−ch2−n+h3
Answers: 3

Chemistry, 22.06.2019 21:20
Phosgene (carbonyl chloride), cocl2, is an extremely toxic gas that is used in manufacturing certain dyes and plastics. phosgene can be produced by reacting carbon monoxide and chlorine gas at high temperatures: co(g) cl2(g)⇌cocl2(g) carbon monoxide and chlorine gas are allowed to react in a sealed vessel at 477 ∘c . at equilibrium, the concentrations were measured and the following results obtained: gas partial pressure (atm) co 0.830 cl2 1.30 cocl2 0.220 what is the equilibrium constant, kp, of this reaction
Answers: 2

You know the right answer?
The pressure inside of a sealed syringe of hydrogen gas is 80.0KPa with a volume of 250 Liters. What...
Questions





Biology, 01.08.2019 13:47




History, 01.08.2019 13:47


History, 01.08.2019 13:47


Mathematics, 01.08.2019 13:47



History, 01.08.2019 13:47



Biology, 01.08.2019 13:47