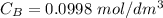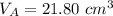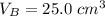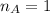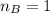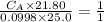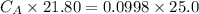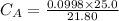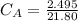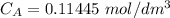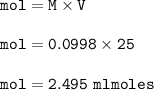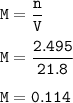Student carries out a titration to determine the concentration of a solution of
nitric acid. She titrates the solution of nitric acid against a standard solution
of sodium hydroxide with a known concentration of 0.0998 mol/dm². She
finds that 21.80 cm of the nitric acid solution is needed to exactly neutralise
25.0 cm of the sodium hydroxide solution.
Calculate the concentration of the nitric acid solution. Give your answer to
three significant figures.
The equation for the neutralisation reaction is
HNO3 + NaOH → NaNO3 + H2O

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:00
24. a sports ball is inflated to an internal pressure of 1.85 atm at room temperature (25 °c). if the ball is then played with outside where the temperature is 7.5 °c, what will be the new pressure of the ball? assume the ball does not change in volume nor does any air leak from the ball a) 0.555 atm b) 1.74 atm c) 1.85 atm d) 1.97 atm
Answers: 2

Chemistry, 22.06.2019 16:00
Answer asap : ( a. how does mucus prevent the entry of pathogens? b. describe two ways white blood cells protect us from pathogens.
Answers: 1

Chemistry, 23.06.2019 01:00
An unsaturated hydrocarbon is a hydrogen-carbon compound with a. a network solid structure b. single bonds c. single bonds in a branched-chain structure d. double or triple bonds
Answers: 1

Chemistry, 23.06.2019 14:00
Which is not true regarding reaction rates? (2 points) catalysts are not used up in the reaction. catalysts speed up reactions by lowering the activation energy. reaction rates decrease as the concentration of reactants decrease. during reactions, concentrations of all reactants decrease at the same rate.
Answers: 1
You know the right answer?
Student carries out a titration to determine the concentration of a solution of
nitric acid. She ti...
Questions

English, 31.01.2021 01:00

English, 31.01.2021 01:00


Health, 31.01.2021 01:00



Mathematics, 31.01.2021 01:00

Mathematics, 31.01.2021 01:00


Social Studies, 31.01.2021 01:00

Mathematics, 31.01.2021 01:00

Mathematics, 31.01.2021 01:00





Computers and Technology, 31.01.2021 01:00


Mathematics, 31.01.2021 01:00

Mathematics, 31.01.2021 01:00

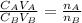
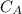 is the concentration of acid
is the concentration of acid
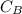 is the concentration of base
is the concentration of base
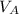 is the volume of acid
is the volume of acid
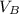 is the volume of base
is the volume of base
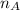 is the mole ratio of acid
is the mole ratio of acid
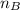 is the mole ratio of base
is the mole ratio of base