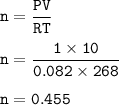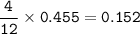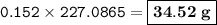Chemistry, 04.12.2020 14:00 littlemrslazy
Suppose that 10.0 L of Carbon Dioxide gas are produced by this reaction, 4C3H5N3O9 -> 12 CO2 + 10H2O + 6N2 +O2, at a temperature of -5 degrees C, and a pressure of exactly 1 atm. Calculate the mass of nitroglycerin that must have reacted in grams.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:10
How many moles of gas are present if p=11 atm, v=12l, t=185k?
Answers: 1

Chemistry, 22.06.2019 12:40
In the following table, all the columns for the element calcium are filled out correctly. element electron structure of atom electron structure of ion net ionic charge calcium 1s22s22p63s23p64s2 1s32s22p63s23p64s1 +1 true false
Answers: 2


Chemistry, 22.06.2019 23:40
What energy conversion occurs when a sling shot is used to shoot a rock across the room? (2 points) question 2 options: 1) stored mechanical energy is converted to mechanical energy. 2) stored mechanical energy is converted to radiant energy. 3) gravitational energy is converted to radiant energy. 4) gravitational energy is converted to mechanical energy.
Answers: 1
You know the right answer?
Suppose that 10.0 L of Carbon Dioxide gas are produced by this reaction, 4C3H5N3O9 -> 12 CO2 + 10...
Questions



History, 27.08.2019 02:40

Social Studies, 27.08.2019 02:40


English, 27.08.2019 02:40

Social Studies, 27.08.2019 02:40


History, 27.08.2019 02:40

Mathematics, 27.08.2019 02:40


Spanish, 27.08.2019 02:40

History, 27.08.2019 02:40

Mathematics, 27.08.2019 02:40

Computers and Technology, 27.08.2019 02:40

Physics, 27.08.2019 02:40

Computers and Technology, 27.08.2019 02:40


History, 27.08.2019 02:40

Mathematics, 27.08.2019 02:40