
Chemistry, 03.11.2020 04:10 michaellevingston46
Can someone help me solve question 'a' please? Just need to know how you did it so I can do the rest.
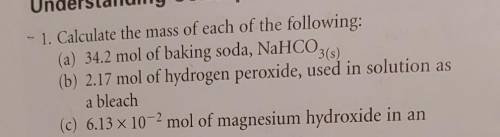

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 17:10
Nitric oxide (no) can be formed from nitrogen, hydrogen and oxygen in two steps. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 2 h_2(g) rightarrow 2 nh_3 (g) delta h = -92. kj in the second step, ammonia and oxygen react to form nitric oxide and water: 4 nh_3(g) + 5 o_2(g) rightarrow 4no(g) + 6 h_2o(g) delta h = -905. kj calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 1


Chemistry, 22.06.2019 02:50
What is the overall order of reaction for rate = k[no]2[o2]
Answers: 3

Chemistry, 23.06.2019 00:30
If there are 3.5 moles of koh, how many moles of naoh can be produced? question 1 options: a)3.0 moles naoh b)3.5 moles naoh c)1 moles naoh d)9 moles naoh
Answers: 1
You know the right answer?
Can someone help me solve question 'a' please? Just need to know how you did it so I can do the rest...
Questions

Geography, 22.10.2019 18:10



Mathematics, 22.10.2019 18:10






Mathematics, 22.10.2019 18:10

Mathematics, 22.10.2019 18:10

Biology, 22.10.2019 18:10


Spanish, 22.10.2019 18:10

Health, 22.10.2019 18:10

Mathematics, 22.10.2019 18:10


Social Studies, 22.10.2019 18:10




