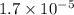Express a mass of 0.017 mg in grams.
Answer in units of g....

Answers: 1
Another question on Chemistry


Chemistry, 23.06.2019 06:40
The combustion of methane, ch4, releases 890.4kj/mol. that is, when one mole of methane is burned,890.4 kj are given off to the surroundings. this meansthat the products have 890.4 kj less than the reactants.thus, ah for the reaction = - 890.4 kj. a negative symbolforah indicates an exothermic reaction.ch (g) + 20 (g)> co2 (g) + 2 h0 (1); ah = - 890.4 kga) how much energy is given off when 2.00 mol of ch,are burned? b) how much energy is released when 22.4g of ch. areburned?
Answers: 1

Chemistry, 23.06.2019 12:10
Which structure is a valid representation of a hydrocarbon molecule?
Answers: 2

Chemistry, 23.06.2019 12:30
17) large amounts of very important metal titanium are made by reacting magnesium metal with titanium tetrachloride. titanium metal and magnesium chloride are produced. a) write the balanced equation for this reaction. b) how many kilograms of magnesium are required to produce 1.00 kilograms of titanium? ( show work, .)
Answers: 1
You know the right answer?
Questions


Mathematics, 14.12.2020 15:10


Mathematics, 14.12.2020 15:10

Mathematics, 14.12.2020 15:10

Geography, 14.12.2020 15:10


Mathematics, 14.12.2020 15:10

Biology, 14.12.2020 15:10


Mathematics, 14.12.2020 15:10

Mathematics, 14.12.2020 15:20




Mathematics, 14.12.2020 15:20


Mathematics, 14.12.2020 15:20

English, 14.12.2020 15:20