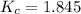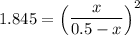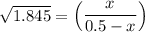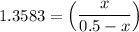Chemistry, 05.09.2020 20:01 Rperez6491
For the reaction:
CO(g) + H2O(g) ⇌ CO2(g) + H2(g)
the value of Kc is 1.845 at a specific temperature. We place 0.500mol CO and 0.500mol H2O in a 1.00L container at this temperature and allow the reaction to reach equilibrium. Determine the equilibrium concentration of all species present in the container.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 13:50
What does standard deviation reveal about data? a. the average of all the data points b. which of the data points is most reliable c. how spread out the data points are d. the percent error included in the data
Answers: 2

Chemistry, 22.06.2019 03:00
Atrain travels 74 kilometers in 3 hours, and then 81 kilometers in 5 hours. what is its average speed?
Answers: 2

Chemistry, 22.06.2019 04:00
Gymnast always perform on padded mats. how does the mats protect the gymnast
Answers: 2

Chemistry, 22.06.2019 05:30
The table describes how some substances were formed substance 19 description formed by boiling pure water formed by combining three hydrogen atoms to every nitrogen atom formed by adding 5 g of sugar to 1 l of water formed by compressing carbon under high pressure based on the given descriptions, which substance is most likely a mixture?
Answers: 1
You know the right answer?
For the reaction:
CO(g) + H2O(g) ⇌ CO2(g) + H2(g)
the value of Kc is 1.845 at a specifi...
the value of Kc is 1.845 at a specifi...
Questions





Business, 20.09.2020 14:01


Social Studies, 20.09.2020 14:01





Mathematics, 20.09.2020 14:01


Biology, 20.09.2020 14:01

World Languages, 20.09.2020 14:01

Mathematics, 20.09.2020 14:01

History, 20.09.2020 14:01

Mathematics, 20.09.2020 14:01

Physics, 20.09.2020 14:01

![K_c = \dfrac{[x][x]}{[0.5-x][0.5-x]}](/tpl/images/0743/5235/b80f7.png)
![K_c = \dfrac{[x]^2}{[0.5-x]^2}](/tpl/images/0743/5235/7f3bb.png)