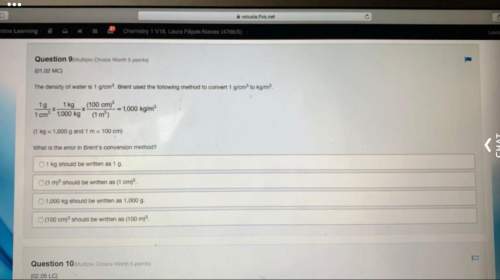
Chemistry, 27.07.2020 06:01 frandariusscott
37.2 liters of a gas has a pressure of 362.43 kPa at 46.5 °C. If the pressure increases to
693.9 kPa and the temperature to 149.2°C, what would be the new volume of the gas?
Select one:
O a. 25.7
O b. 62.3
O c. 54
O d. 25.672

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 18:30
What volume of a 0.0606 m solution of strontium bromide is needed to obtain 0.340 mol of the compound? question 42 options: a)5.61 l b) 3.4 l c) 600 ml d) 1 l e) 178 ml
Answers: 1

Chemistry, 22.06.2019 21:30
Isopropyl alcohol, (ch3)2choh, is a common solvent. determine the percent by mass of hydrogen in isopropyl alcohol. a) 6.71% h b) 13.4% h c) 25.0% h d) 53.3% h
Answers: 1

Chemistry, 22.06.2019 23:30
If maltose undergoes hydrolysis what subunits does it results to?
Answers: 2

Chemistry, 23.06.2019 00:30
An ice cube with a volume of 45.0ml and a density of 0.9000g/cm3 floats in a liquid with a density of 1.36g/ml. what volume of the cube is submerged in the liquid
Answers: 3
You know the right answer?
37.2 liters of a gas has a pressure of 362.43 kPa at 46.5 °C. If the pressure increases to
693.9 kP...
Questions





Mathematics, 13.01.2020 07:31



Mathematics, 13.01.2020 07:31

Mathematics, 13.01.2020 07:31


Chemistry, 13.01.2020 07:31


Mathematics, 13.01.2020 07:31


Mathematics, 13.01.2020 07:31




Mathematics, 13.01.2020 07:31

Mathematics, 13.01.2020 07:31