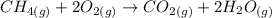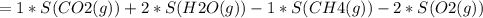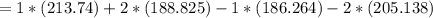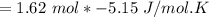Chemistry, 27.06.2020 18:01 destiny3200
Consider the reaction CH4(g) 2O2(g)CO2(g) 2H2O(g) Using standard thermodynamic data at 298K, calculate the entropy change for the surroundings when 1.62 moles of CH4(g) react at standard conditions.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 12:30
Write the chemical formula for a compound that is made of an element from group 1 and an element from group 17
Answers: 1

Chemistry, 22.06.2019 18:10
Given the following at 25c calculate delta hf for hcn (g) at 25c. 2nh3 (g) +3o2 (g) + 2ch4 (g) > 2hcn (g) + 6h2o (g) delta h rxn= -870.8 kj. delta hf=-80.3 kj/mol for nh3 (g), -74.6 kj/mol for ch4, and -241.8 kj/mol for h2o (g)
Answers: 1

Chemistry, 22.06.2019 21:50
What is a main difference between a mixture and a pure substance? a mixture is only a liquid, but a pure substance can be in any state.a mixture looks the same throughout, but a pure substance does not.1 a mixture can vary in composition, but a pure substance has a set composlo a mixture can be made up of a single compound, but a pure substance car
Answers: 2

You know the right answer?
Consider the reaction CH4(g) 2O2(g)CO2(g) 2H2O(g) Using standard thermodynamic data at 298K, calcula...
Questions

Social Studies, 31.07.2019 19:00







Social Studies, 31.07.2019 19:00

Mathematics, 31.07.2019 19:00

History, 31.07.2019 19:00

Mathematics, 31.07.2019 19:00

Biology, 31.07.2019 19:00



Mathematics, 31.07.2019 19:00

English, 31.07.2019 19:00


History, 31.07.2019 19:00

Mathematics, 31.07.2019 19:00