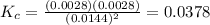Chemistry, 27.06.2020 15:01 cynthiafchs9203
Given the reaction: 2HF(g) H2(g) F2(g) If the initial concentration of HF is 0.025M and the equilibrium concentration of H2 is 0.0028M, then what is the equilibrium constant of the reaction

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:30
Explain why pure hydrogen cyanide does not conduct electricity, but become a conductor when it is dissolved in water? (at room temp, pure hcn exists as a volatile liquid)
Answers: 1

Chemistry, 22.06.2019 14:50
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 23:10
Using the periodic table, complete the following. element: hydrogen symbol: h₂ molecular weight: g mass of one mole: g/mol
Answers: 3

Chemistry, 23.06.2019 05:50
Aseismic wave is energy released as the result of rock movement along a fault. t or f ?
Answers: 1
You know the right answer?
Given the reaction: 2HF(g) H2(g) F2(g) If the initial concentration of HF is 0.025M and the equilibr...
Questions



English, 03.12.2020 20:40

Mathematics, 03.12.2020 20:40

History, 03.12.2020 20:40

Mathematics, 03.12.2020 20:40


Social Studies, 03.12.2020 20:40

Mathematics, 03.12.2020 20:40


Mathematics, 03.12.2020 20:40




Advanced Placement (AP), 03.12.2020 20:40

Physics, 03.12.2020 20:40



Business, 03.12.2020 20:40

![K_{c} =\frac{[H_{2}][F_{2}] }{[HF]^2}](/tpl/images/0695/8873/96c9f.png)