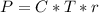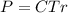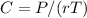Chemistry, 11.06.2020 16:02 bethdove9466
The osmotic pressure exerted by a solution is equal to the molarity multiplied by the absolute temperature and the gas constant . Suppose the osmotic pressure of a certain solution is measured to be at an absolute temperature o of 312. K. Write an equation that will let you calculate the molarity c of this solution.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 18:30
Covalent network solids typically have melting points and boiling points. the chemical formula of a network solid indicates in the molecule.
Answers: 3

Chemistry, 22.06.2019 13:50
What happens when an atom of sulfur combines with two atoms of chlorine to produce sci2? a. each chlorine atom shares a pair of electrons with the sulfur atom. b. an electron is transferred from each chlorine atom to the sulfur atom. c. an electron is transferred from the sulfur atom to each chlorine atom. d. each chlorine atom shares all its valence electrons with the sulfur atom.
Answers: 2

Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2

Chemistry, 22.06.2019 20:30
Identify the correct mole ratio for each substance. sodium chloride (nacl) na: cl = 1: ammonium nitrate (nhno) h: o = 4:
Answers: 1
You know the right answer?
The osmotic pressure exerted by a solution is equal to the molarity multiplied by the absolute tempe...
Questions


English, 10.02.2021 03:30

Mathematics, 10.02.2021 03:30

Business, 10.02.2021 03:30

Mathematics, 10.02.2021 03:30


Mathematics, 10.02.2021 03:30

Mathematics, 10.02.2021 03:30

Mathematics, 10.02.2021 03:30



Mathematics, 10.02.2021 03:30

Mathematics, 10.02.2021 03:30


Mathematics, 10.02.2021 03:30



Mathematics, 10.02.2021 03:30